Properties of Electric Charge is a key and high-scoring topic in Class 12 Physics Electrostatics. For CBSE and RBSE board exams, it's important to clearly understand concepts like conservation, quantization, and additivity of charge, as questions are often asked from this section. In this article, you will get simple explanations, important points, and quick revision notes to help you score better.
Introduction to Electric Charge
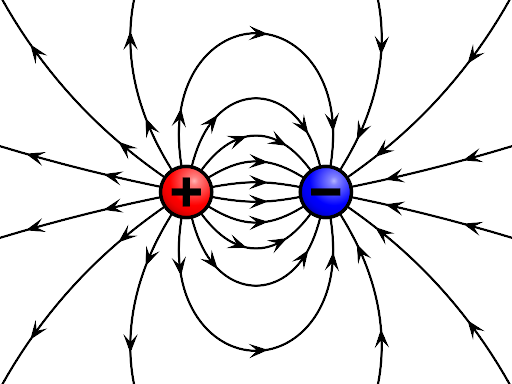 |
| Electric Charge |
Electric charge is a fundamental property of matter that causes it to experience a force when placed in an electromagnetic field. It exists in two types: positive and negative. Like charges repel each other, while unlike charges attract. This interaction is explained by Coulomb's Law, which describes the force between charged objects. Electric charge also follows key principles such as conservation and quantization. These properties are based on electrostatics and play a crucial role in understanding various natural phenomena and modern technologies used in everyday life.
 |
| Like and Unlike Charges |
What is Electric Charge
Key details about electric charge:
- Fundamental Property: Electric charge is an inherent property of subatomic particles. Protons carry a positive charge, while electrons carry a negative charge, forming the basis of all electrical phenomena.
- Attraction and Repulsion: In electrostatics, like charges repel each other, whereas opposite charges attract, which explains many everyday electrical interactions.
- Conservation and Transfer: According to the law of conservation of charge, electric charge cannot be created or destroyed; it can only be transferred from one object to another.
- Quantization: Electric charge exists in discrete amounts and follows the principle of quantization of charge, given by
- Types of Charge: A body becomes positively charged when it loses electrons and negatively charged when it gains electrons.
- Units: The SI unit of electric charge is the Coulomb (C). One Coulomb is equal to the charge of approximately `6.25 \times 10^{18}C` elementary charges.
Types of Electric Charge
- Positive Charge (+): A deficiency of electrons is called a positive charge; it means the object has more protons than electrons.
- Negative Charge (-): An excess of electrons is called a negative charge; it means the object has more electrons than protons.
- Neutral (0): An object is neutral when the number of protons and electrons is equal, so that the total charge is zero.
SI Unit of Electric Charge
- Definition of Quantization of Charge
Key Aspects of Charge Quantization
- Formula of Quantization of Charge:
- Origin: This comes from the fact that electric charge is transferred in the form of whole electrons during processes such as charging by friction, so charge always appears in discrete (quantized) amounts.
- Discovery: The idea was first indicated by Michael Faraday through his electrolysis experiments, and later experimentally confirmed by Robert Millikan in 1912.
- Macroscopic Scale: At the macroscopic scale, charge is quantized but appears continuous because the number of elementary charges is extremely large, making individual charge units practically unnoticeable.
- Example:
- Charge always comes in packets of elementary charge (e).
- Just like you cannot have half a person, you cannot have half an electron's charge.
- That's why n must always be an integer.
Key Principles
- Net Charge Invariance: The algebraic sum of all positive and negative charges in a closed system always remains constant.
- Transfer vs. Creation: Charging occurs due to the movement of electrons. If an object becomes positively charged, it has lost electrons, which are gained by another object.
- Universal Application: This law holds true in all situations, from everyday static electricity to advanced nuclear and particle physics processes.
Examples in Nature
- Static Electricity
- Radioactive Decay
- Pair Production/Annihilation
Key Concepts
- Isolated System: The law of Conservation of Charge only applies if no charge enters or leaves the system.
- Scalar Quantity: Charge is a scalar quantity; charges are added algebraically, considering their signs (+q or - q).
- Role of Electrons: Charge transfer occurs exclusively through the movement of electrons, not protons.
- Mass association: Charge is a property of particles like electrons and protons which have mass.
- Algebraic Sum: Since electric charges can be either positive or negative, their signs must be taken into account while adding them.
- Scalar Nature: Electric charge is a scalar quantity (it has magnitude but no direction), which is why we can add them like simple numbers.
- Formula:
- Example:
- Charges simply add like numbers (taking signs into account).
- This follows the principle of Conservation of Charge - charge is neither created nor destroyed, only transferred.
Invariance of Electric Charge
Conclusion
Properties of Electric Charges MCQs
1. What is the SI unit of electric charge?
a) Joule
b) Ampere
c) Coulomb
d) Volt
2. Which subatomic particle carries a negative electric charge?
a) Proton
b) Neutron
c) Electron
d) Positron
3. What happens when two objects with the same charge are brought close together?
a) They attract each other.
b) They repel each other.
c) They remain stationary.
d) None of the above.
4. What is the term for the process of transferring electric charge by direct contact?
a) Induction
b) Conduction
c) Radiation
d) Insulation
5. Which law describes the force between two charged objects?
a) Newton's Law
b) Kepler's Law
c) Coulomb's Law
d) Faraday's Law
6. What is the charge of a neutron?
a) Positive
b) Negative
c) Neutral
d) Variable
Short Answer Type Questions
1. Define electric charge and explain its two types.
2. How do conductors differ from insulators concerning the movement of electric charge?
3. Explain why a charged object attracts a neutral object.
4. How does lightning occur, and what role does electric charge play in the process?
5. Differentiate between direct current (DC) and alternating current (AC).
6. How does the concept of electric charge apply to the function of electronic devices?
Answer MCQs
1. What is the SI unit of electric charge?
c) Coulomb
2. Which subatomic particle carries a negative electric charge?
c) Electron
3. Which of the following materials is an insulator?
b) Rubber
4. What happens when two objects with the same charge are brought close together?
b) They repel each other.
5. What happens when a positively charged object is brought near a neutral object?
b) The neutral object gains a negative charge.
6. Which of the following is a unit of electric potential difference?
b) Volt
7. What is the term for the process of transferring electric charge by direct contact?
b) Conduction
8. Which law describes the force between two charged objects?
c) Coulomb's Law
9. What is the charge of a neutron?
c) Neutral
10. Which type of current flows in only one direction?
b) Direct current
Call to Action (CTA)
If you found this post helpful, don't stop here-take your preparation to the next level with structured resources and guided learning.
- Free Important Question PDF Download
- Join Physics365 WhatsApp-Get daily questions & updates. Join Now
- Watch Full Video- Physics 365


No comments:
New comments are not allowed.